OrthoLogic Corporation (NASDAQ:CAPS) is a little unusual for us. While it trades below its net cash value and Biotechnology Value Fund (BVF) has disclosed a 13.42% holding, BVF’s holding is passive. At CAPS’s $0.60 close yesterday it has a market capitalization of $24.4M. We estimate the net cash value to be 80% higher at $1.08 per share. CAPS’s cash burn rate is quite high relative to its net cash position, so rapid steps need to be taken for this to be a profitable investment. We think that BVF is a good bet, so we’re adding CAPS to the Greenbackd Portfolio.
About CAPS
CAPS is a development stage biotechnology company focused on the development and commercialization of the synthetic peptides Chrysalin (TP508) and AZX100. Effective October 1, 2008, OrthoLogic Corp. is known and doing business as Capstone Therapeutics. The company’s investor relations website is here.
The value proposition
The summary of our estimate for the company’s liquidation value is set out below (the “Book Value” column shows the assets as they are carried in the financial statements, and the “Liquidating Value” column shows our estimate of the value of the assets in a liquidation):
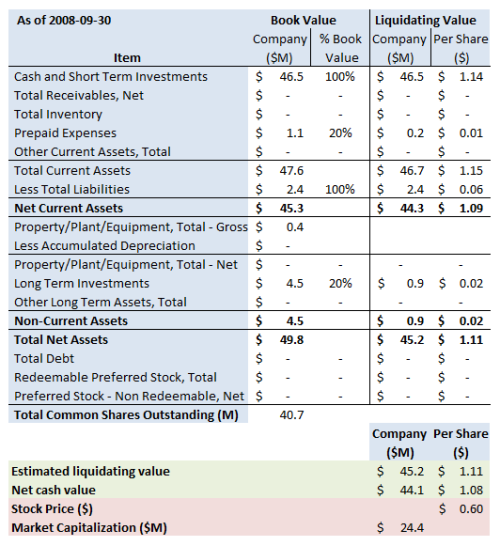
Note that we have used the September 10Q. CAPS’s most recent filings indicate that cash is actually $48M (see slide 13), but we don’t know what the rest of the balance sheet looks like. The presentation also gives cash burn guidance this year of $14M to $16M.
The company also included the following in its 10Q, which seems to indicate a shift to cash preservation:
We announced that we have no immediate plans to re-enter clinical trials for Chrysalin-based product candidates and a strategic shift in our development approach to our Chrysalin Product Platform. We currently intend to pursue development partnering or licensing opportunities for our Chrysalin-based product candidates, a change from our previous development history of independently conducting human clinical trials necessary to advance our Chrysalin-based product candidates to market. We will continue to explore Chrysalin’s therapeutic value in tissues and diseases exhibiting endothelial dysfunction as well as the science behind and potential of Chrysalin. We will also continue research and development expenditures for further pre-clinical studies supporting multiple indications for AZX100 and plan to continue AZX100 dermal scarring human clinical trials.
Off-balance sheet arrangements and Contractual obligations
There is no discussion in the September 10Q about CAPS’s off-balance sheet arrangements or contractual obligations.
The catalyst
Given that BVF has filed a 13G notice, which indicates a passive investment, we’re not entirely sure what BVF has planned for CAPS. It’s possible that it is simply a passive holding. We’re reasonably comfortable following BVF into CAPS because of their efforts with Avigen Inc (NASDAQ:AVGN) and Neurobiological Technologies Inc (NASDAQ:NTII).
CAPS has been undertaking a stock repurchase program since March 5, 2008. At September 30, 2008, the company had repurchased 1.1.M shares of its common stock, at a total cost of $1.0M, and had allocated approximately a further $1.1M to fund possible future stock repurchases. We don’t know the status of the buy-back at this time.
Conclusion
At its $0.60 close yesterday, CAPS is trading at 55% of our estimate of its $1.08 per share net cash value. The risk for this investment – as it is for all of these types of investment – is that CAPS dissipates its cash before it or BVF can salvage that value. Management is taking steps to reduce its cash burn and repurchase undervalued stock, which is encouraging. Perhaps this is what BVF has seen, and the reason BVF hasn’t filed a 13D notice. We think that BVF is a good bet, so we’re adding CAPS to the Greenbackd Portfolio.
CAPS closed yesterday at $0.60.
The S&P500 Index closed yesterday at 752.83.
Hat tip to ef.
[Full Disclosure: We do not have a holding in CAPS. This is neither a recommendation to buy or sell any securities. All information provided believed to be reliable and presented for information purposes only. Do your own research before investing in any security.]


G-
Thanks for your write up on CAPS.
The off balance sheet liabilities are minimal – – an operating lease with a total commitment of $1 mm or $0.03 per share.
It may very well be that BVF is in this to eventually call for a liquidation. However, the AZX100 drug, which CAPS is developing could be a home run (but I am not a biotech guy so no clue on the merits of the drug).
Take a look at what Shire paid to Renovo for a similar drug which is still under development (see press release below).
The preliminary results from 50% of the patients enrolled in the AZX100 Phase II will be released in early 2010. So the way to look at this investment is that by the end of 2010, there should be approximately 70-75 cents per share of liquidation value left if the drug failed. Tack on some cost to liquidate and you will still come up with higher value than where the stock is trading. In the mean time, you get a free option to ride on in AZX100 and Chrysalin might have some residual value as well (but I wouldnt count on it…..)
Press Release:
Shire pays $825m to develop Renovo’s scar-reduction remedy
By Karen Attwood
Thursday, 21 June 2007
Share
Shire is to pay up to $825m (£414m) to Renovo for the exclusive development and commercialisation rights to the biopharmaceutical company’s groundbreaking scar-prevention drug.
Shares in Renovo jumped 14 per cent, up 27p to 214p, as the City welcomed the significantly higher-than-expected deal for the drug. Juvista was developed after experiments that started 20 years ago revealed that alligator embryos displayed exceptional wound-healing qualities.
Shire will pay $75m cash upfront and will also take a $50m stake in the AIM-listed company, equivalent to about 7 per cent of the equity. Further payments will be triggered as specific milestones are reached, such as FDA approval and sales targets being met. Shares in Shire were off a penny at 1220p.
Owing to the wide range of scars the drug could treat, from serious medical conditions such as burns through to routine surgery and cosmetic surgery, analysts say Juvista has blockbuster potential with peak sales estimated at $1.2bn.
The potential market for preventative scar treatment in the US alone is believed to be about $4bn with 42 million people undergoing surgery each year, according to Renovo. Some 90 per cent of surgeons would use a preventative drug if it were on the market, according to research, while the majority of patients say they would be prepared to pay for anti-scar treatment. At present there is nothing in development that is promising the same kind of treatment as Juvista.
Renovo’s chief executive, Mark Ferguson, said the company has completed trials on more than 1,500 people. Results have been “very positive”, he said. Juvista is moving into final stage trials in the middle of next year and the drug could be launched by 2011.
Samir Devani, an analyst at Nomura Code, said the terms of the deal were “outstanding”. “Critically, Renovo has retained European marketing rights and we expect the company to now develop its own sales and marketing structure,” he added.
The deal marks a departure for Shire, the UK’s third biggest drug-maker, which is focused on therapeutic medicines such as its best-selling attention deficit hyperactivity disorder drug, Adderall XR. Analysts said it fitted the company’s speciality niche focus, while Goldman Sachs pointed out that Shire has the opportunity to build a franchise in cosmetic and general surgery application with Juvista in a similar way to how Allergan grew its franchise with Botox.
LikeLike
Thanks, Double F. We very much appreciate your comments here.
G
LikeLike